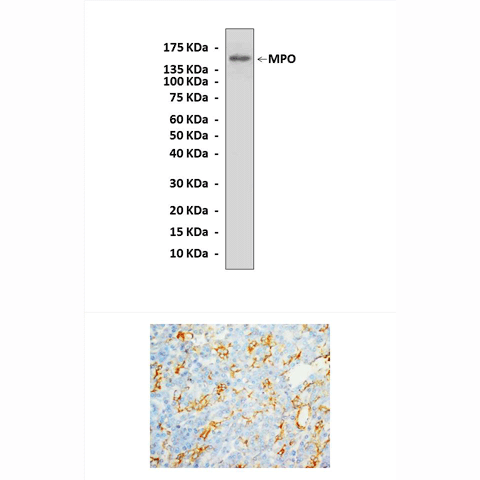
Anti-MPO: Rabbit Myeloperoxidase Antibody |
 |
BACKGROUND Myeloperoxidase (MPO) is a heme protein abundantly expressed in polymorphonuclear neutrophils (PMNs), and accounting for up to 5% of total cell protein. It plays an important role in innate immunity and regulation of the generation of nitric oxide (NO)-derived oxidants. MPO exists as a 150-kDa tetramer composed of two glycosylated 59- to 64-kDa heavy chain and two nonglycosylated 14-kDa light chains. MPO is released from activated PMNs into the phagosome and extracellular space, where the enzyme utilizes H2O2 from the respiratory burst to catalyze the formation of hypochlorous acid (HOCl) and variety of other oxidizing species. HOCl displays bactericidal activity in vitro.1 However recent observations expand this view and show that MPO-derived oxidants are critically involved in a more subtle modulation of signaling pathways. For example, low levels of MPO-derived hypochlorous acid has been demonstrated to activate mitogen-activated protein (MAP) kinases, induce nuclear translocation of transcription factors, regulate cell growth by activating tumor-suppressor proteins, or modulate the activity of metalloproteinases. Also, vascular cell glycosaminoglycan-associated MPO and MPO-derived free radical intermediates interfere with vascular signaling pathways by oxidizing endothelial derived nitric oxide (NO). Interestingly, MPO not only proved to adhere to endothelial cells but has also been implicated in PMN membrane association. It was shown that MPO binds to CD11b/CD18 integrins on PMNs, leading to induction of intracellular signaling cascades and translating into up-regulated PMN degranulation, CD11b surface expression, and NADPH oxidase activity in an autocrine manner.2
MPO activity measurement is used as an index of inflammation, because the MPO levels are increased in inflammatory tissue largely secondary to PMN extravasation. MPO is also present in human atherosclerotic lesions, and plasma MPO activity may be an excellent marker of acute coronary syndromes. The MPO-induced catalysis of low-density lipoprotein oxidation converts low-density lipoprotein into its high uptake form and thus contributes to the formation of foam cells and cholesterol deposition in the vessel wall. MPO activates the formation of protein nitrotyrosine (NO2Tyr), a characteristic of acute and chronic inflammation. In addition, The MPO was localized with fibronectin in the extracellular matrix and induced tissue remodeling by nitration of matrix proteins.3
MPO activity measurement is used as an index of inflammation, because the MPO levels are increased in inflammatory tissue largely secondary to PMN extravasation. MPO is also present in human atherosclerotic lesions, and plasma MPO activity may be an excellent marker of acute coronary syndromes. The MPO-induced catalysis of low-density lipoprotein oxidation converts low-density lipoprotein into its high uptake form and thus contributes to the formation of foam cells and cholesterol deposition in the vessel wall. MPO activates the formation of protein nitrotyrosine (NO2Tyr), a characteristic of acute and chronic inflammation. In addition, The MPO was localized with fibronectin in the extracellular matrix and induced tissue remodeling by nitration of matrix proteins.3
REFERENCES
1. Guilpain, P. et al: Autoimmun. Rev. 7:421-5, 2008
2. Lau, D. et al: Proc. Natl. Acad. Sci. USA 102:431-6, 2005
3. Tiruppathi, C. et al: Proc. Natl. Acad. Sci. USA 101:7699-704, 2004
2. Lau, D. et al: Proc. Natl. Acad. Sci. USA 102:431-6, 2005
3. Tiruppathi, C. et al: Proc. Natl. Acad. Sci. USA 101:7699-704, 2004
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CA1054 |
Antigen: | Short peptide from human MPO sequence. |
Isotype: | Rabbit IgG |
Species & predicted species cross- reactivity ( ): | Human, Rat |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC 1:50 - 1:200 ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 150 kDa |
Specificity/Sensitivity: | Detects endogenous levels of MPO proteins without cross-reactivity with other related proteins. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой